Finwaver.com
Your school or business runs better on finwaver.com. Sign up for free one (1) week try.
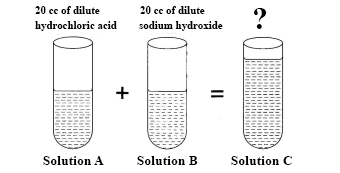
In an experiment, equal volumes of dilute hydrochloric acid (Solution A) and dilute sodium hydroxide (Solution B) are mixed together to form solution C.
(i)
What is the volume of solution C?
(ii)
Red litmus paper and blue litmus paper are dipped in turns into solutions A, B and C.
State the observations you will make in all six cases.
(iii)
Give the name of the reaction that took place between solution A and solution B.
(iv)
Identify solution C
(v)
State what will happen when solution C is heated.
In an experiment, red and blue litmus papers were dipped separately into three test tubes containing one of the test substances listed in the table below.
| Test substances | Observation | Conclusion | |
| Red litmus paper | Blue litmus paper | ||
| Lemon juice | |||
| Calcium hydroxide | |||
| Dilute hydrochloric acid | |||
(i)
Copy and complete the table by making the necessary observation and conclusion for each substance.
(ii)
Name two of the test substances that would react with each other to produce salt and water.
(iii)
Write down a balanced chemical equation for the reaction in (ii) above.
In an experiment to investigate the reactivity of zinc, a piece of the metal was dropped into a test tube containing dilute hydrochloric acid. The experiment set-up is illustrated below.
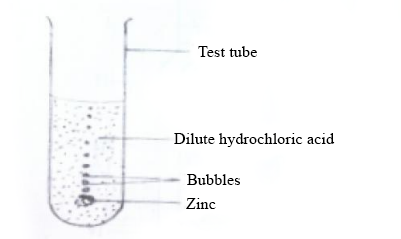
Study the set-up carefully and answer the questions that follow.
(i)
Write a balanced chemical equation for the reaction that occurred in the experiment.
(ii)
Name the gas evolved.
(iii)
List two metals which can react in a similar way as the zinc.
(iv)
List two metals which cannot react in a similar way as the zinc.
(v)
Name two glass apparatus which could have been used instead of the test tube.
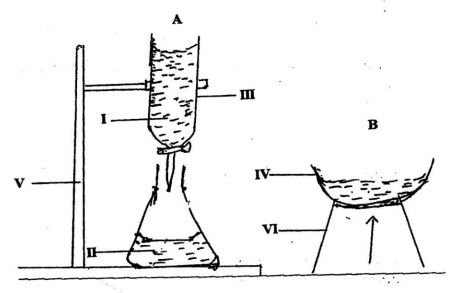
The diagrams below is a set-up for preparation of common salt in the laboratory. Study the diagrams carefully and answer the questions that follow.
(i)
Name each of the parts labelled IV, V and VI.
(ii)
Name two possible solutions that can react to product salt.
(iii)
Name the process that takes place when the two solution named in (ii) react.
(iv)
Name the process that takes place in the set-up B.
(v)
Write a balanced chemical equation for the reaction between the two solutions named in (ii).
In an experiment, equal volumes and equal concentrations of dilute hydrochloric acid and dilute sodium hydroxide solutions were each placed in different test tubes.
Read the following statements carefully.
I. Both red and blue litmus papers were dipped into each of the solutions in turns.
II. Equal volumes of the solutions were mixed to obtain a third solution.
III. Both red and blue litmus papers were dipped into the third solution.
Use the information provided to answer the following questions.
i)
Explain briefly how you can identify each of the solutions
α)
Hydrochloric acid
β)
Sodium hydroxide
ii)
State the types of reaction that occurred when the two solutions were mixed.
iii)
What type of solution was formed when the reaction stated in (ii) occurred?
iv)
State what would be observed when both red and blue litmus papers were dipped into the third solution.
v)
Explain how the solid portion of the solution named in (iii) could be obtained.