Finwaver.com
Your school or business runs better on finwaver.com. Sign up for free one (1) week try.
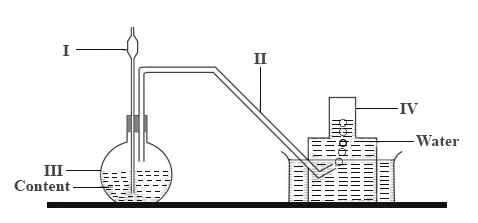
The set-up below is used to prepare gases in the laboratory.
(i)
Give the names of the parts labelled I, II, III and IV.
(ii)
Write down the two names of the method of gas collection.
(iii)
Explain how the gas collects over the water.
(iv)
Name two gases that can be prepared using the set-up.
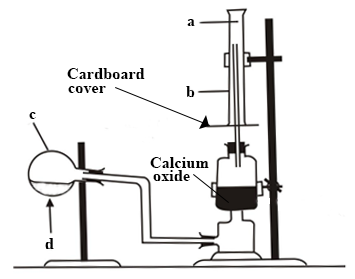
The set-up below is used in the preparation of ammonia gas in the laboratory
Study it and answer the questions that follow.
(i)
Name the parts labelled a, b and c
(ii)
What is the meaning of the arrow sign d?
(iii)
What is the function of the calcium oxide?
(iv)
Why has c been tilted downwards?
(v)
State the method by which the gas is collected.
(vi)
How will you test for the gas?
(vii)
Give the names and the chemical formulae of the compounds that form the content of c.
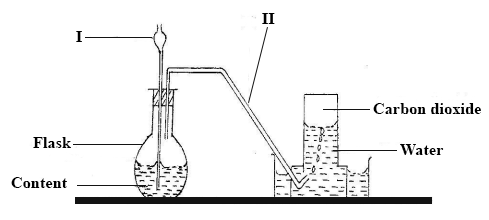
The diagram below shows the set-up for the preparation of carbon dioxide in the laboratory.
(i)
Name the parts of the set-up labelled I and II.
(ii)
Give the name of the method of gas collection shown in the diagram.
(iii)
Give one property of gases collected over water.
(iv)
What will happen if component I does not dip into the contents of the flask?
(v)
List two compounds that form the content of the flask.
(vi)
Write down the systematic name of carbon dioxide.