1.
The IUPAC name of the compound whose structural formula is illustrated below is
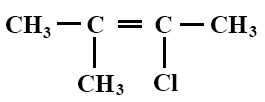
2-chloro-3-methylbut-2-ene.
2-methyl-2-chlorobut-2-ene.
2-chloro-3-methyl-2-butane.
2-methyl-2-chloro-2-butyne.
2.
Organic compounds are mostly
soluble in water.
not inflammable.
covalent.
ionic.
3.
Which of the following compounds is a hydrocarbon?
C5H8
C6H12O6
CH3CH2OH
CH3COCH3
4.
Ash from burnt plants is widely used in Ghana
as bleaching powder.
for preparing soap.
for preparing salt.
for improving soil texture.
5.
The manufacture of plastics involves the scientific process known as
crystallization.
fermentation.
vulcanization.
Polymerization.
6.
Which of the following combinations of elements do all carbohydrates contain?
Sulphur, carbon, hydrogen and oxygen
Carbon, phosphorus, oxygen and hydrogen
Carbon, hydrogen and nitrogen
Carbon, hydrogen and oxygen
7.
The gas used in the welding torch belongs to the family of
alkanoates.
alkanes.
alkynes.
alkenes.
8.
The fraction of petroleum distillation used as fuel for heavy duty engine is
bitumen.
diesel.
gasoline.
kerosene.
9.
The IUPAC name of the compound whose structural formula is illustrated below is
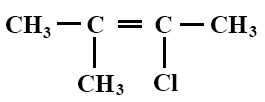
2-chloro-3-methylbut-2-ene.
2-methyl-2-chlorobut-2-ene.
2-chloro-3-methyl-2-butane.
2-methyl-2-chloro-2-butyne.
10.
Organic compounds are mostly
soluble in water.
not inflammable.
covalent.
ionic.
11.
Which of the following compounds of the electromagnetic spectrum has the highest frequency?
Gamma rays
Infra-red radiations
Microwave
Ultraviolet radiations
12.
Which of the following compounds is a hydrocarbon?
C5H8
C6H12O6
CH3CH2OH
CH3COCH3
13.
Fehling's solution is used in the laboratory to test for the presence of
fats.
starch.
reducing sugars.
fatty acids.
14.
The substance formed when carbon (IV) oxide s bubbled through lime water for a long time is
Ca(HCO3)2.
Na2CO3.
NaHCO3.
CaCO3.
15.
Which of the following combinations of elements do all carbohydrates contain?
Sulphur, carbon, hydrogen and oxygen
Carbon, phosphorus, oxygen and hydrogen
Carbon, hydrogen and nitrogen
Carbon, hydrogen and oxygen
16.
The component of air used for welding is
O2.
N2.
CO2.
C2H2.
17.
The gas used in the welding torch belongs to the family of
alkanoates.
alkanes.
alkynes.
alkenes.
18.
The fraction of petroleum distillation used as fuel for heavy duty engine is
bitumen.
diesel.
gasoline.
kerosene.
19.
Alkanoic acid + alkanol ⇋H2SO4 alkyl alkanoate + water
Use the equation above to answer the question below
The reaction is called
hydrolysis.
hydration.
dehydration.
esterification.
20.
Alkanoic acid + alkanol ⇋H2SO4 alkyl alkanoate + water
Use the equation above to answer the question below
The H2SO4 is acting as
catalyst.
enzyme.
drying agent
dehydrating agent.
21.
Which of the following properties of carbon dioxide is useful in fire fighting?
I. It does not support combustion.
II. It is heavier than air.
III. It does not produce toxic compounds.
I and II only
I and III only
II and III only
I, II and III
22.
The general formula of alkyne is
CnH2n+2
CnH2n
CnH2n-2
CnHn
23.
The reaction represented by the equation
RCOOH + R'OH ⇋ RCOOR' + H2O is an example of
esterification.
hydrolysis.
neutralization.
oxidation.
24.
The IUPAC name of the compound Mg(HCO3)2 is
magnesium hydrogen trioxocarbonate (III).
magnesium hydrogen trioxocarbonate (IV).
magnesium hydrogen trioxocarbonate (V).
magnesium hydrogen trioxocarbonate (II).
25.
How many moles of HCl is contained in 14.6 g of HCl? [H = 1, Cl = 35.5]
0.4 mol
0.04 mol
0.004 mol
4.0 mol
26.
The functional group for alkanol is
-C=C-
C=C
-C-C-
-OH
27.
Which of the following chemical equations is balanced?
Na + Cl₂ → NaCl
Mg + O₂ → MgO
K + O₂ → K₂O
2Na + Cl₂ → 2NaCl
28.
Chemistry of carbon compounds is studied under
biochemistry.
inorganic Chemistry.
organic Chemistry.
physical Chemistry.