1.
A body has a mass of 70 kg. What is the force due to the earth acting on it? [g = 10 ms-2]
3500 N
700 N
70 N
7000 N
2.
The main practice which is carried out in the nursery just before seedlings are transplanted is
shading
pricking-out
hardening-off
thinning
3.
The diagram below is an electrical circuit showing two resistors connected in parallel. Determine the effective resistance of the combination.
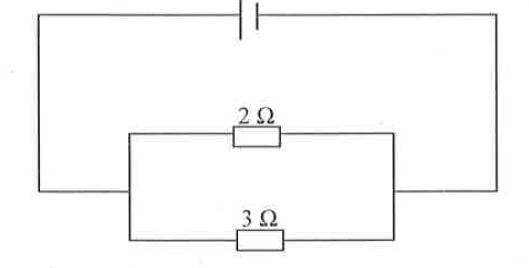
1.20 Ω
0.83 Ω
5.00 Ω
6.00 Ω
4.
The following statements are examples of application of biotechnology in agriculture except
improving upon colour and size of a flower
development of pest-resistant crops
crossing animals with desired characteristics
introduction of foreign genes into livestock
5.
The movement of water into root hairs is by
osmosis
diffusion
capillarity
transpiration
6.
An example of soil micronutrient is
nitrogen
iron
iron
sulphur
7.
Which of the following systems in the human body is directly affected when carbon monoxide is inhaled?
Respiratory
Excretory
Circulatory
Circulatory
8.
The part of the human brain that controls body temperature is
medulla oblongata
hypothalamus
cortex
cerebellum
9.
How many moles of HCl is contained in 14.6 g of HCl? [H = 1, Cl = 35.5]
0.4 mol
0.04 mol
0.004 mol
4.0 mol
10.
In which of the following blood vessels is the pressure highest?
Dorsal aorta
Renal artery
Hepatic artery
Vena cava
11.
The functional group for alkanol is
-C=C-
C=C
-C-C-
-OH
12.
Which of the following statements about magnetic fields are correct? A magnetic field
I. has magnetic lines of force.
II. is a force field.
III. has direction.
II and III only
I and III only
I and II only
I, II and III
13.
Substances excreted by the skin of humans include
salts and water only
urea and salts only
water and urea only
salts, urea and water only
14.
The type of livestock raised in any area depends mainly on the
inputs available
availability of land
type of climate
availability of labour
15.
The IUPAC name for Na₂SO₄ is
sodium tetraoxosulphate (IV)
disodium sulphate
sodium (II) sulphate
sodium tetraoxosulphate (VI)
16.
Which of the following vegetables provide the largest amount of roughage in the diet of humans?
Carrot
Cabbage
Beans
Pepper
17.
The immunization of children is important because it
increases the production of white blood cells
stimulates the production of antibodies
destroys pathogens in the body
stimulates the production of antigens
18.
What is the kinetic energy of a body of mass 20 kg moving with a velocity of 4 m s-1?
80 J
160 J
320 J
5 J
19.
The gas produced when a dilute hydrochloric acid reacts with calcium carbonate is
chlorine
carbon dioxide
carbon monoxide
hydrogen
20.
Which of the following statements about the image formed in a pinhole camera are correct? The image is
I. inverted.
II. upright.
III. real.
II and III only
I and III only
I and II only
I and II only
21.
Which of the following traits in humans is an example of continuous variation?
Body mass
Colour blindness
Gender
Blood group
22.
Which of the following chemical equations is balanced?
Na + Cl₂ → NaCl
Mg + O₂ → MgO
K + O₂ → K₂O
2Na + Cl₂ → 2NaCl
23.
The instruments used to measure the mass of a body include
I. top pan balance.
II. spring balance.
III. electronic balance.
II and III only
I and III only
I and III only
I and II only
24.
Which of the following animals is a non-ruminant herbivore?
Poultry
Pig
Cattle
Rabbit
25.
The energy needed for natural cycles to operate is obtained from
fossils
the sun
the air
decomposers
26.
Ectoparasites of farm animals are effectively controlled by
handpicking
drenching
dipping
quarantine
27.
A device which converts chemical energy to electrical energy is
resistor
cell
bulb
voltmeter
28.
A flowering plant with poorly developed roots, yellow buds and yellowing new leaves is likely to be deficient in
manganese
copper
boron
sulphur
29.
Water is not suitable as a thermometric liquid because it
I. is opaque.
II. wets glass.
III. expands abnormally.
II and III only
I and III only
I and II only
I, II and III
30.
Conversion of nitrites to nitrates in the nitrogen cycle is caused by
Azotobacter
Nitrobacter
Nitrosomonas
Rhizobium
31.
In the control of malaria, the main aim of spraying stagnant water bodies is to
ensure that the adult mosquitoes have no hiding place
prevent the larvae from breathing in air
make the water unsuitable for egg development
prevent the laying of eggs by the mosquitoes
32.
Aluminium does not corrode easily because it
forms aluminium oxide on its surface
is a strong metal
is hard
forms aluminium hydroxide on its surface
33.
Which part of the leaf is the amount of glucose produced during photosynthesis the least?
Guard cell
Palisade mesophyll
Spongy mesophyll
Upper epidermis
34.
Which of the following physical properties of water are correct?
I. It is tasteless.
II. It has a pH of 7.
III. It is colourless.
II and III only
I and III only
I and II only
I, II and III
35.
During rusting, iron reacts with oxygen to form
iron (III) oxide
iron (IV) oxide
iron (II) oxide
iron (V) oxide
36.
Members of the same species living at the same place at the same time make a/an
population
ecosystem
community
trophic level
37.
The westerlies are winds that blow
from the poles
towards the poles
towards the equator
from the equator
38.
The P-N junction diode has
equal number of holes and electrons
no holes
no electrons
more electrons than holes
39.
The correct sequence of the arrangement of vertebrae in a mammal is
thoracic, caudal, lumbar, cervical
cervical, lumbar, thoracic, caudal
thoracic, cervical, lumbar, caudal
cervical, thoracic, lumbar, caudal
40.
Which of the following structures is present in animal cells only?
Mitochondrion
Lysosomes
Golgi body
Ribosome
41.
Loss of appetite and poor growth in humans are due to the deficiency of
copper
zinc
iron
iodine
42.
Nucleons refer to the sum of
electrons and neutrons in an atom
protons and neutrons in an atom
protons and electrons in an atom
electrons and molecules in an atom
43.
Which of the following statements are correct?
I. Greenhouse effect can be reduced by afforestation.
II. One effect of ozone layer depletion is global warming.
III. Greenhouse effect is reduced by bush burning.
II and III only
I and III only
I and II only
I, II and III
44.
The nuclide ²³⁰Th undergoes alpha decay. The mass number and atomic number of the daughter nuclide are respectively
230, 90
226, 88
226, 89
222, 86
45.
Ash from cocoa pod tastes bitter because it contains
alcohol
alkali
acid
soap
46.
A musical note is sound with a definite
intensity
frequency
amplitude
velocity
47.
Hydrological cycle is the system by which water continuously circulates through the
troposphere
biosphere
atmosphere
mesosphere
48.
The structure associated with the absorption of fatty acids and glycerol in the small intestines is the
lacteals of the villi
lymphatic system
blood capillaries
hepatic portal veins
49.
It is most difficult to detect carbon (II) oxide gas than most poisonous gases because it
does not support combustion
is insoluble in water
is heavier than air
has no smell and colour
50.
Metamorphic rocks are formed through
chemical change caused by heat and pressure
biological change of original rocks
physical disintegration of igneous rocks
combination of igneous and sedimentary rocks
(a)
(i)
What are exotic breeds?
[2 marks]
(ii)
State three advantages of exotic breeds of poultry.
[2 marks]
(b)
(i)
Define relative atomic mass.
[3 marks]
(ii)
Calculate the molar mass of each of the following compounds:
(α) Na₂CO₃
(β) CH₃COOH
[Na = 23, O = 16, C = 12, H = 1]
[2 marks]
(c)
(i)
Name two organisms commonly used in biotechnology.
[2 marks]
(ii)
Give one example of an industry which uses each of the organisms named in (i).
[2 marks]
(d)
(i)
List three other sources of electric power generation apart from hydro and thermal sources.
[2 marks]
(ii)
State the energy transformation that takes place in any two of the listed sources.
[2 marks]
(a)
(i)
The nuclides of sodium and fluorine are represented as 2311Na and 199F respectively. State the number of:
(α) protons in each atom;
(β) neutrons in each atom.
[2 marks]
(ii)
Draw a diagram to show the electron configuration of:
(α) sodium ion;
(β) fluorine ion.
[4 marks]
(b)
(i)
State two reasons why salt is important in the diet of humans.
[2 marks]
(ii)
Give two examples of an abiotic factor which is non-climatic in terrestrial ecosystem.
[2 marks]
(c)
(i)
What is an electrical conductor?
[2 marks]
(ii)
Two lamps of resistances 3Ω and 5Ω are connected in parallel to a 6V source. Calculate the value of the current supplied by the source.
[3 marks]
(d)
(i)
Distinguish between ear-notching and ear-tagging in farm animals.
[2 marks]
(ii)
List three ecto-parasites that infest goats in West Africa.
[3 marks]
(a)
(i)
State three functions of the respiratory system of humans.
[3 marks]
(ii)
Name one reagent used to test for each of the following food substances in the school laboratory:
(α) proteins;
(β) lipids;
(γ) reducing sugars.
[3 marks]
(b)
(i)
What is a longitudinal wave?
[2 marks]
(ii)
State two differences between music and noise.
[2 marks]
(c)
(i)
Explain the term ruminants as used in animal production.
[2 marks]
(ii)
State three advantages of rearing rabbits.
[3 marks]
(d)
(i)
What is a standard solution?
[2 marks]
(ii)
A standard hydrochloric acid solution is to be prepared from a stock solution of HCl of concentration 10 mol dm⁻³.
(α) What volume of the stock solution is needed to prepare 0.50 dm³ of 2.0 M HCl?
(β) Calculate the volume of water required.
[3 marks]
(a)
(i)
State three advantages that parallel electrical wiring has over series electrical wiring in a household.
[3 marks]
(ii)
An immersion heater is rated 220 V, 850 W. Determine the current flowing through the heater.
[2 marks]
(b)
(i)
Explain the term soil conservation.
[2 marks]
(ii)
State two ways by which each of the following activities help in conserving the soil:
(α) addition of organic matter;
(β) crop rotation.
[4 marks]
(c)
(i)
What are petrochemicals?
[2 marks]
(ii)
Name two uses of petrochemicals in Agriculture.
[2 marks]
(d)
With the aid of a genetic diagram, explain briefly how a couple of normal skin colors produces an albino.
[5 marks]
(a)
(i)
Describe briefly each of the following post-harvest practices:
(i) Winnowing;
(ii) Dehusking;
(iii) Shelling.
[6 marks]
(b)
(i)
Name the type of movable joint found at each of the following locations in the human body:
(α) fingers;
(β) where the neck joins the head.
[2 marks]
(ii)
State one characteristic feature each of the joints named in (i).
[2 marks]
(c)
(i)
What is a magnetic field?
[2 marks]
(ii)
Draw a bar magnet and show the magnetic field around it.
[3 marks]
(d)
(i)
Complete the following chemical equations:
(α) H₂SO₄ + NaOH → ______ + ______
(β) CH₃COOH + CH₃OH → ______ + ______
[3 marks]
(ii)
Name each of the reactions in (i) (a) and (b).
[2 marks]
(a)
State one significance each of the following stages of treatment of water for public consumption:
(i) Aeration;
(ii) Sedimentation;
(iii) Filtration;
(iv) Chlorination;
(v) Coagulation.
[5 marks]
(b)
(i)
What is weight of a body?
[2 marks]
(ii)
The mass of a body is 70 kg. Determine the weight of the body.
[g = 10 m s⁻²]
[3 marks]
(c)
State five challenges associated with pig production in Ghana.
[5 marks]
(d)
(i)
State three effects of Sexually Transmitted Infections (STIs) on the male reproductive system of humans.
[3 marks]
(ii)
Name two glands of the human reproductive system that produce hormones.
[2 marks]